What is the Size of a Proton?
Introduction to Protons
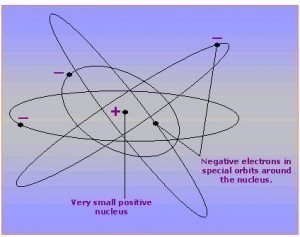 A proton is classified as a subatomic particle. It is part of an atom, located in the nucleus together with the neutron. A proton has a positive electrical charge. The subatomic particle “orbiting” the nucleus, the electron, has a negative charge. A proton’s antiparticle is known as the antiproton. While very small in size, protons are not the tiniest particles in nature. Each proton is made up of quarks, which cannot be divided into smaller particles.
A proton is classified as a subatomic particle. It is part of an atom, located in the nucleus together with the neutron. A proton has a positive electrical charge. The subatomic particle “orbiting” the nucleus, the electron, has a negative charge. A proton’s antiparticle is known as the antiproton. While very small in size, protons are not the tiniest particles in nature. Each proton is made up of quarks, which cannot be divided into smaller particles.
What is the Size of a Proton?
A proton is a very small part of a whole atom. The diameter of a proton is given as 10^ -15m. This is far too small for the human mind to imagine, let alone see. So to comprehend the size of a proton relative to an atom, you will have to scale up the atom first. Say you were to scale an atom, which includes the electron(s) orbiting the nucleus, to 50 meters. The nucleus in its center would be about ½ inch in size. Out of that, a single proton would amount to only half a millimeter. From this you can see that a single atom has lots of “free space” within itself and isn’t a single dot like one can imagine.
What is the Mass Size of a Proton?
In terms of mass, one proton measures 1.672621637 ( 83 )× 10 − 27 kilograms. It is heavier than an electron.
What is the Volume of a Proton?
It is difficult to make an exact measure of a proton’s volume. Some estimates give its diameter as about 2.350 cubic picometers. It is probably smaller than this figure.
More on Protons
Like neutrons, protons are nucleons. Both are attracted by the nuclear force to form the center or nucleus of an atom. Most of an atom’s weight comes from protons and neutrons. Very little is from electrons, the negatively charged particles that orbit at the atom’s periphery.
It used to be thought that protons were elementary particles. An elementary particle cannot be divided into smaller particles. However scientists have discovered that protons are in fact made of “quarks. Each proton consists of three quarks, two are “up” and one is “down.”
What is the Atomic Number?
The atomic number is used to identify a chemical element in chemistry. The atomic number is simply the number of protons in an atom’s nucleus. If two atoms have the same number of protons in their center, they are the same chemical element.